1. Wash 100 mg of amine particles 3 times with 0.1-mM sodium phosphate, pH 7.0 (activation buffer).
2. After the final wash, suspend the particles in 10 ml of coupling buffer containing 0.5% glutaraldehyde (for monomeric activation) or 15% glutaraldehyde (for dimeric activation) and mix well to create a homogeneous suspension.
3. React with mixing for 1 h (for monomeric) or 15 h (for dimeric) at room temperature.
4. Wash the particles with coupling buffer at least several times using centrifugation to remove excess glutaraldehyde. Resuspend in 5 ml of coupling buffer (25-mM sodium phosphate, pH 7.0). Note: The activated particles can be stored at 4°C in water until used.
5. Add the protein to be coupled to the particle suspension in an amount equal to 1- to 10-times molar excess over the calculated particle monolayer for the protein type to be coupled. Mix thoroughly to dissolve. Low concentrations of protein may result in particle aggregation, because a single protein molecule can react with more than one particle.
6. React with mixing at room temperature for 2 to 4 h.
7. Add to the particle suspension a final concentration of 0.2-M glycine (or another amine-containing quench molecule, such as ethanolamine or Tris). The blocking agent will couple to any remaining glutaraldehyde reactive sites. Excess aldehyde groups on the support also may be block by the addition of 50-mM hydroxylamine, which will react with the aldehydes to create stable oxime linkages.
8. Remove excess protein and reactants by washing with coupling buffer at least three times using centrifugation. Storage of the modified particles may be done at 4°C in a suitable buffer containing a preservative.
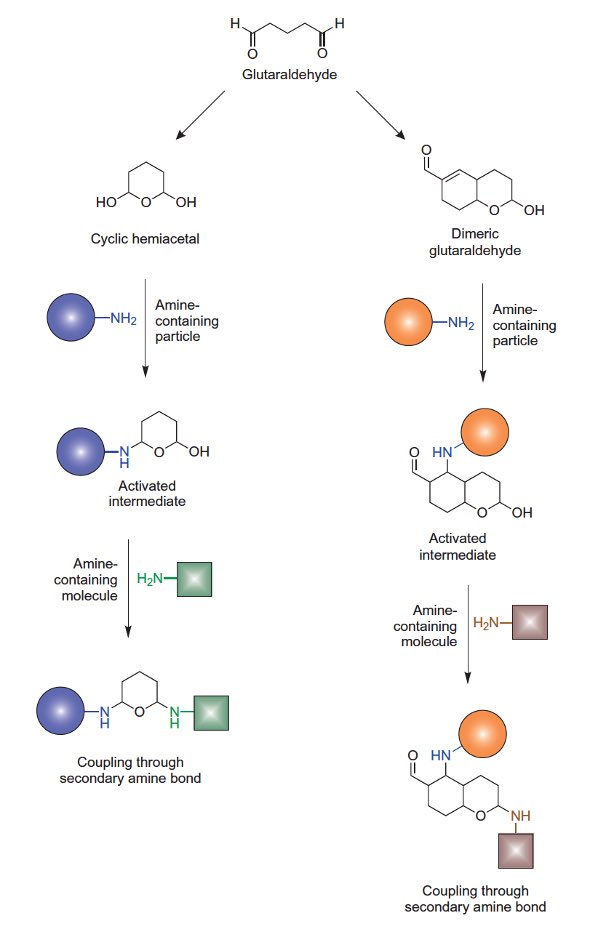
Glutaraldehyde-mediated immobilization onto amine particles can be performed with some control using two main reaction routes. The use of a cyclic hemiacetal glutaraldehyde species in aqueous solution can be used to activate the amino groups by coupling with one of the anomeric carbons on the ring and forming a reactive intermediate. After washing off excess reagent, the activated particles can be used to immobilize an amine-containing protein or ligand through secondary amine linkages. A similar reaction scheme can be carried out using a dimeric glutaraldehyde species that first reacts with the amine groups on particles by addition to the double bond. The final amine-containing ligand then is immobilized through a secondary amine bond to the anomeric carbon of the hemiacetal ring.
Interested in learning more about our particles with versatile functional groups for your conjugation? Visit Our Functional Polystyrene Particles to explore more.
Reference: Bioconjugate Techniques, 3rd Edition - July 25, 2013, Greg T. Hermanson
