Introduction
Carboxylate particles can be coupled to amine-containing molecules using a number of reaction strategies. The most frequently used method involves an aqueous two-step coupling process using EDC and NHS or sulfo-NHS to form an amide bond with a protein or other molecules.
This protocol describes a general method for covalently conjugating amine-containing biomolecules (e.g., proteins, antibodies, oligonucleotides) to carboxylated particles, utilizing EDC (1-Ethyl-3-[3-dimethylaminopropyl]carbodiimide) and Sulfo-NHS (N-hydroxysulfosuccinimide) chemistry.

Figure. Carboxylate particles can be coupled to amine-containing molecules using a number of reaction strategies.
Two-Step EDC/Sulfo-NHS Coupling Protocol
- EDC activates carboxyl groups, which then react with Sulfo-NHS to form NHS esters, creating a more stable and reactive intermediate for coupling to primary amines on the biomolecule.
- This two-step method is preferred for biomolecules containing both amine and carboxyl groups to minimize polymerization.
- Optimization is generally required to achieve optimal activity, stability, and minimal non-specific binding.
I. Materials
- Carboxylated Particles: Microparticles or nanoparticles with carboxylate functional groups.
- Target Biomolecule: Protein, antibody, peptide, oligonucleotide, or other amine-containing ligand.
- Activation Buffer: 50 mM MES (2-(N-morpholino)ethanesulfonic acid), pH 6.0.
- Coupling Buffer: Phosphate-buffered saline (PBS), pH 7.2-8.5.
- EDC (1-Ethyl-3-[3-dimethylaminopropyl]carbodiimide): Water-soluble carbodiimide.
- Sulfo-NHS (N-hydroxysulfosuccinimide): Enhances EDC coupling efficiency.
- Quenching Solution: Ethanolamine, Tris, glycine, or other amine-containing molecule.
- Washing Buffer: PBS with a mild detergent such as 0.05% Tween-20.
- Storage Buffer: Appropriate buffer for long-term storage of the bioconjugate, often containing a preservative.
- Deionized Water
- Microcentrifuge tubes
- Centrifuge
- Vortex mixer
- Rotator or shaker
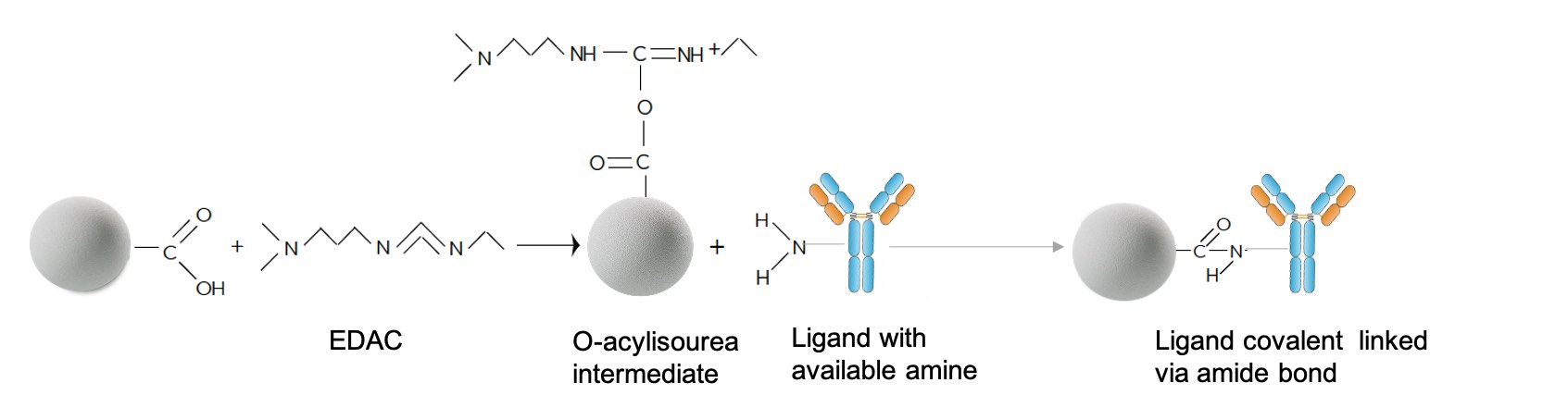
Figure. Carboxyl-modified microspheres can be conjugated to a biomolecule using two-step reaction
II. Protocol
A. Particle Preparation
1. Wash Carboxylate Particles: Wash the particles with coupling buffer or deionized water using centrifugation or magnetic separation (if applicable).
- Purpose: To remove any storage buffer, preservatives, or unbound substances.
Note: Avoid buffers containing carboxylates or amines (e.g., acetate, glycine, Tris, imidazole) during washing.
2. Resuspend Particles: Resuspend the washed particles in activation buffer at a suitable concentration (e.g., 10 mg/mL).
- Optional: Add a dilute detergent (e.g., 0.01% SDS) to increase particle stability and prevent clumping.
B. Activation of Carboxylate Groups
1. Prepare EDC and Sulfo-NHS Solutions: Dissolve EDC and Sulfo-NHS separately in activation buffer immediately before use.
Reason: EDC is prone to hydrolysis, so use fresh solutions.
2. Mix EDC/Sulfo-NHS and Add to Particles: Combine the EDC and Sulfo-NHS solutions, then add this mixture to the particle suspension.
- Typical Concentrations: Optimize for your specific application.
3. React: Incubate the mixture at room temperature for 15-30 minutes with gentle mixing.
- Mechanism: EDC activates carboxyl groups, and Sulfo-NHS enhances the reaction with amines.
C. Biomolecule Coupling
1. Wash Activated Particles: Remove excess EDC and Sulfo-NHS by washing the particles 2-3 times with coupling buffer.
- Method: Use centrifugation or magnetic separation, as appropriate.
2. Prepare Biomolecule Solution: Dissolve the biomolecule in coupling buffer at the desired concentration.
- Concentration Range: Typically, a 1- to 10-fold molar excess of ligand over the maximal calculated carboxylate group concentration is recommended.
3. Combine Biomolecule and Particles: Add the biomolecule solution to the activated particle suspension.
4. React: Incubate the mixture at room temperature or 4°C for 2-4 hours or overnight with gentle mixing or rotation.
D. Quenching and Blocking
1. Quench Unreacted Sites: Add a quenching solution (e.g., ethanolamine, Tris, or glycine) to block any remaining active sites on the particles.
- Concentration: Use a concentration (e.g., 100 mM ethanolamine or 0.2 M glycine) sufficient to block excess reactive sites.
- Incubate: Mix for 30 minutes to 1 hour at room temperature.
2. Wash the Bioconjugate: Remove excess quenching agent and unbound biomolecule by washing the particles multiple times with washing buffer.
E. Storage
1. Resuspend the Bioconjugate: Resuspend the particles in an appropriate storage buffer containing a preservative.
- Example: PBS with 0.02% sodium azide or another suitable preservative.
2. Store the Bioconjugate: Store at 4°C or according to the manufacturer’s instructions.
III. Key Considerations and Optimization
- pH: Maintain the correct pH for activation and coupling.
- Ligand Concentration: Optimize the ligand concentration to achieve desired surface coverage without aggregation.
- Reaction Time and Temperature: Adjust the reaction time and temperature to optimize conjugation efficiency while preserving biomolecule activity.
- Washing: Ensure thorough washing to remove unreacted reagents and prevent nonspecific binding.
- Quenching: Use appropriate quenching agents to block unreacted sites.
- Stability: Add stabilizers (e.g., BSA) to prevent aggregation and maintain activity.
- Solvent Compatibility: Confirm particle stability in the buffers and solvents used.
- Controls: Run proper controls for specificity and to ensure proper results.
- Nanoparticle considerations: Take into account that conjugation reactions performed with nanoparticles may have greater reactivity than those with microparticles. Re-optimize the protocol when using nanoparticles.
- Buffer Considerations: The compatibility of the buffer and ligand (solubility, activity) is important, and should be considered when selecting buffers.
- Literature Search: Prior to beginning, perform a thorough literature search for immobilization protocols specific to your biomolecule of interest.
- Alternative buffers: Alternative coupling buffers for immobilization onto NHS-activated supports include 0.1-M MOPS (pH 7.0), 0.1- to 0.2-M phosphate (pH 7.2–7.5), 0.1 to 0.2-M NaHCO3 (pH 8.0), or 0.1-M sodium borate (pH 8.5), which all may or may not contain NaCl.
Interested in learning more about our particles with versatile functional groups for your conjugation? Visit Our Functional Polystyrene Particles to explore more.
IV. Safety Precautions
- Wear appropriate PPE, including gloves, lab coat, and eye protection.
- Handle EDC and Sulfo-NHS in a fume hood to avoid inhalation.
- Dispose of chemical waste properly according to local regulations.
Reference: Bioconjugate Techniques, 3rd Edition - July 25, 2013, Greg T. Hermanson
