I. Introduction
This protocol outlines a general method for covalently conjugating biomolecules to amine-modified beads. It uses a homobifunctional crosslinker, such as glutaraldehyde, imidoesters, or NHS esters, to link available amine groups on a ligand to the amine-functionalized beads.
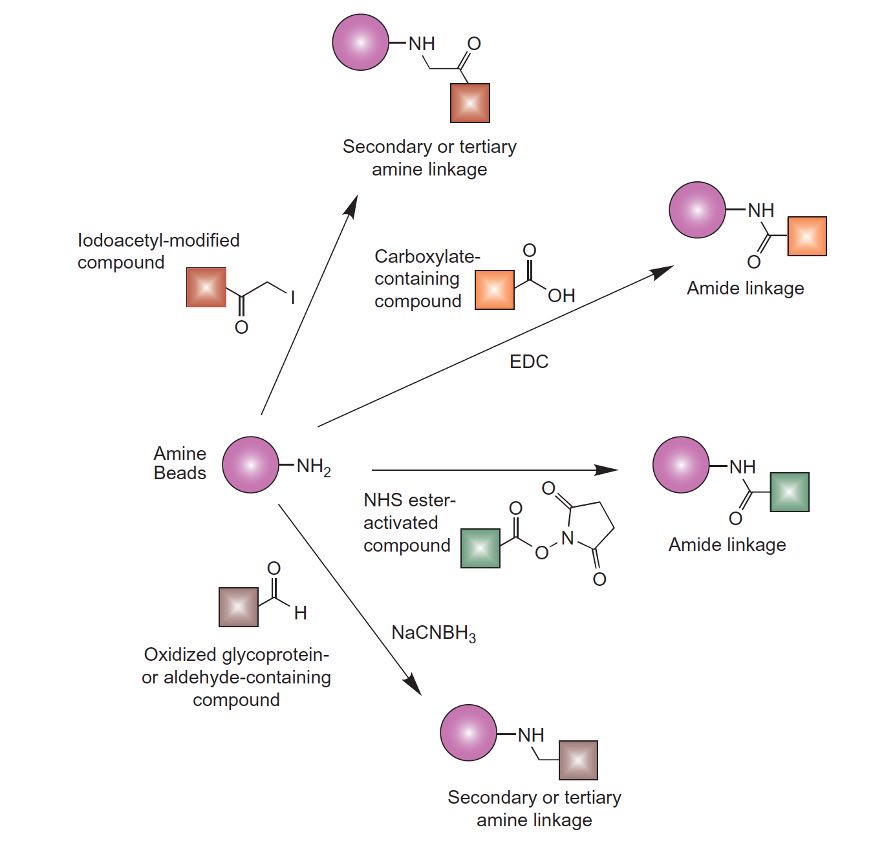 Figure.Amine-containing particles can be conjugated using alkylation or acylation reactions to result in secondary or tertiary amine linkages or amide bonds.
Figure.Amine-containing particles can be conjugated using alkylation or acylation reactions to result in secondary or tertiary amine linkages or amide bonds.
II. Materials
- Amino-modified microspheres: Often supplied as a 10% solids solution.
- Amine-reactive homobifunctional crosslinker: e.g., glutaraldehyde, imidoesters, or NHS esters.
- Wash/coupling buffer: pH 6.0-9.0.
- Target biomolecule: Protein, antibody, peptide, oligonucleotide, or other amine-containing ligand.
- Quenching solution: With a primary amine source, 30-40 mM [e.g., hydroxylamine, ethanolamine, glycine, etc.].
- Blocking molecule: 0.05-1% (w/v).
- Storage buffer: pH 7.0-7.5, with 0.01-0.1% (w/v) blocking molecule.
- Deionized Water
- Microcentrifuge tubes
- Centrifuge
- Vortex mixer
- Rotator or shaker
III. Protocol
1. Wash the Amino-Modified Microspheres:
- Wash the microspheres two times using a wash/coupling buffer.
- Use centrifugation or magnetic separation (if applicable) to separate the beads after each wash.
- Purpose: To remove any storage buffer, preservatives, or unbound substances.
2. Activate Microspheres with Crosslinker:
- Resuspend the washed microspheres in the wash/coupling buffer. Ensure the microspheres are well suspended by vortexing, sonicating, or rolling.
- Add the amine-reactive homobifunctional crosslinker (e.g., glutaraldehyde) to the microsphere suspension.
- Incubate the mixture at room temperature for the duration recommended by the crosslinker's manufacturer.
- Note: For glutaraldehyde activation, Betancor et al. (2006) procedures can be followed. Activation can be performed using any chromatographic support material containing available primary amines. The final level of glutaraldehyde activation can be controlled by regulating the amount of these amines produced initially on the support material.
3. Couple the Target Biomolecule:
- Wash the activated microspheres with the wash/coupling buffer to remove any unreacted crosslinker.
- Resuspend the microspheres in the coupling buffer containing the protein or other biomolecule of interest.
- Incubate the mixture at room temperature or 4°C for 2-4 hours or overnight with gentle mixing or rotation.
4. Quench Unreacted Sites:
- Add a quenching solution (e.g., hydroxylamine, ethanolamine, or glycine) to block any remaining active sites on the microspheres.
- Incubate for 30 minutes to 1 hour at room temperature.
5. Wash the Bioconjugate:
- Remove excess quenching agent and unbound biomolecule by washing the particles multiple times with washing buffer.
6. Storage:
- Resuspend the particles in an appropriate storage buffer, pH 7.0-7.5, containing a blocking molecule.
- Store at 4°C or according to the manufacturer’s instructions.
IV. Considerations and Optimization
- pH: Maintain the correct pH for activation and coupling using the wash/coupling buffer.
- Crosslinker Concentration: Optimize the crosslinker concentration to achieve the desired surface coverage without causing excessive crosslinking or aggregation.
- Ligand Concentration: Optimize the ligand concentration to achieve desired surface coverage without aggregation.
- Reaction Time and Temperature: Adjust the reaction time and temperature to optimize conjugation efficiency while preserving biomolecule activity.
- Washing: Ensure thorough washing to remove unreacted reagents and prevent nonspecific binding.
- Quenching: Use appropriate quenching agents to block unreacted sites.
- Stability: Add stabilizers (e.g., BSA) to prevent aggregation and maintain activity.
- Literature Search: Prior to beginning, perform a thorough literature search for immobilization protocols specific to your biomolecule of interest.
- Buffer: Recipes for common buffers can be found in Tech Support Doc 0300, Buffers. Ready to use buffers and solutions are also available.
V. Safety Precautions
- Wear appropriate PPE, including gloves, lab coat, and eye protection.
- Handle crosslinkers in a fume hood to avoid inhalation.
- Dispose of chemical waste properly according to local regulations.
Interested in learning more about our particles with versatile functional groups for your conjugation? Visit Our Functional Polystyrene Particles to explore more.
Reference: Bioconjugate Techniques, 3rd Edition - July 25, 2013, Greg T. Hermanson
